 Because of this, these ions or atoms lack a particular shape. Ionic atomic radius definition: This is the atomic radius that forms an ionic bond. 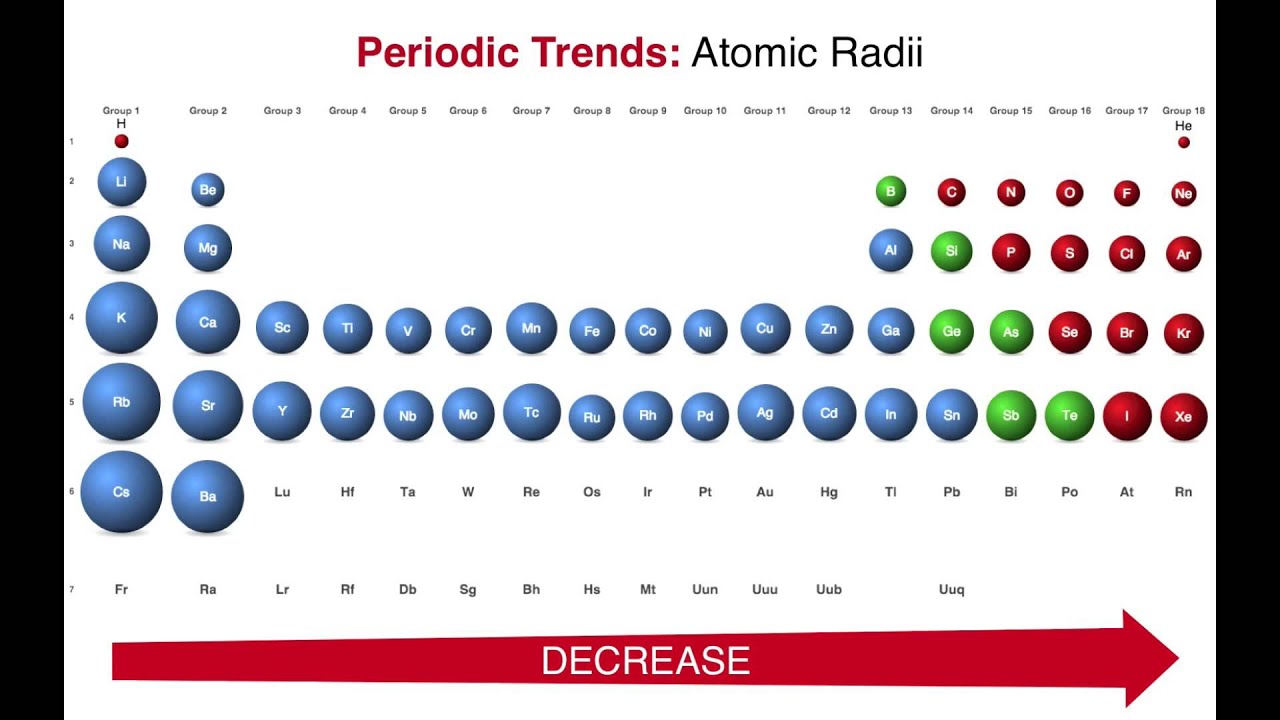
While this is straightforward for some molecules like CI2 and O2, in some cases, the covalent radius needs to be inferred by measuring the bond lengths of atoms with already known radii. If the two atoms in the pair are similar, the covalent radius will equal half of the bond’s total length. It can be determined by measuring the lengths between the pairs of atoms that are covalently bonded. Types of Atomic Radius Concerning the Types of Bondĭepending on the type of bond an atom forms, atomic radii are divided into the following three types:Ĭovalent atomic radius definition: This is the radius of an atom that is measured when the atom is covalently bonded with another atom of a similar element. In general, the sodium atomic radius is 227 pm, while the aluminium atomic radius is 143 pm. So, an atom has no fixed radius, and the radii depend upon the type of bonds the atoms form. Atomic radii are different depending on the type of bond an atom forms. Finding atomic radius is difficult as there is uncertainty about the position of the outermost orbit of electrons.Ī precise measurement of the atomic radius can be obtained by using Heisenberg’s Uncertainty Principle, according to which the radius is determined based on the distance between two atoms bonded to each other. Atomic radius can be compared to the radius of a circle, where the nucleus corresponds to the centre of the circle, while the outermost orbit can be compared to the outer edge of the circle. The atomic radius of a chemical element is defined as the average distance from the atom’s nucleus to the boundary of the shells of electrons surrounding it. /PeriodicTable_AtomSizes-56a131193df78cf772684720.png)
The atomic radius definition: Atomic radius can be defined as the total distance between the centre of the atomic nucleus to the outermost orbit its electrons are revolving in. This blog is focused on the discussion of atomic radius and its characteristics. Atomic radius refers to the total distance from the centre of the nucleus to the orbit that lies at the greatest distance from the nucleus. At the same time, the neutrons and protons lie inside an area at the centre of the atom known as the nucleus the electrons orbit around this nucleus in a circular path. An atom comprises three main subatomic particles: electrons, neutrons, and protons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
